Marco Mangone
-
Phone: 480-965-7957
-
-
BDA 220F Tempe, AZ 85287-6401
-
Mail code: 6401Campus: Tempe
-
Marco Mangone joined the faculty of ASU in August 2011 after completing his postdoctoral fellowship in the Center for Genomics and Systems Biology at New York University. He is an assistant professor in the Biodesign Virginia G. Piper Center for Personalized Diagnotics and the School of Life Sciences at ASU.
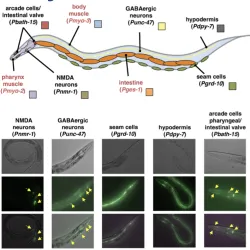
He is interested in study how eukaryotic RNA transcription is terminated and how the messenger RNA is regulated on its way to the expression into proteins, using the wound worm C. elegans as model system. C. elegans is a transparent, free-living soil nematode that shares many biological similarities with humans, and has been used to model many aspects of human biology, such as development, learning and memory, mechanisms of aging and cell death.
His research focuses to the analysis of 3'Untranslated Regions (3'UTRs); sequences located between the STOP codon and the polyA tail of mature messenger RNAs. 3'UTRs contain elements targeted by many non-coding RNAs and proteins that bind and repress the translation of the mRNA into protein. The majority of these elements are unknown, but they have been found to play key roles in diverse developmental and metabolic processes, and are implicated in disease, including diabetes, Alzheimer's and cancer.
Recent analysis of genome-wide data in human, mouse, worms, plants and yeast showed that alternative polyadenylation (APA), a mechanism in which the same gene has multiple 3'UTR isoforms, is pervasive in eukaryotes. It is not known why eukaryotic mRNAs require so many 3'UTR isoforms. Professor Mangone suspects that APA provides a powerful regulatory mechanism, perhaps to regulate gene expression in a tissue-specific manner or in different developmental contests.
Professor Mangone's approach combines high-throughput genomics, bioinformatics, genetics, biochemistry and systems biology to answer the following fundamental question: How does the termination of transcription work in eukaryotes? What are the mechanisms behind alternative polyadenylation? Why is it so widespread in metazoan transcriptomes? How does it impact gene regulation at a post-transcriptional level? What are the mechanisms of 3'end formation that ultimately determine how a gene is regulated?
- Ph.D. Molecular Biology, Watson School of Biological Sciences. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY, USA Advisor: Dr. Winship Herr. Thesis: “Analysis of the HCF–1 basic region and its role in sustaining cell proliferation” 2000–2006
- Dottorato Italian Laurea, La Sapienza University, Rome, Italy. Advisor: Dr. Ernesto Di Mauro. Thesis: “Computational analysis of Single–Nucleotide Polymorphisms in humans” 1990–2000
Dr. Mangone lab uses standard molecular biology and bioinformatics to study the production, function, and disease contribution of 3’untranslated regions (3’UTRs) and their role in modulating gene expression, taking advantage of both C. elegans and human genomics. Please use this link for more information.
Peer–Reviewed Publications generated from work completely conducted at ASU as ASU faculty
2017
- Blazie S, Geissel H, Wilky H, Joshi R, Newbern J and Mangone M (2017) Alternative polyadenylation directs tissue specific miRNA targeting in Caenorhabditis elegans somatic tissues. GENETICS (2017) June 1, vol. 206 no. 2 757-774
- Commentary: Khraiwesh B and Salehi-Ashtiani K Alternative Poly(A) Tails Meet miRNA Targeting in Caenorhabditis elegans. GENETICS (2017) June 1 vol. 206 no. 2 755-756
- Wolter JM, Le HH, Linse A, Godlove VA, Nguyen TD, Kotagama K, Lynch A, Rawls A and Mangone M (2017) Evolutionary patterns of metazoan microRNAs reveal targeting principles in the let-7 and miR-10 families. Genome Res. (2017) 27: 53-63
2016
- Hutchins E.D., Eckalbar W.L., Wolter J.M., Mangone M., Kusumi K. Differential expression of conserved and novel microRNAs during tail regeneration in the lizard Anolis carolinensis. BMC Genomics. 2016 May 5;17:3392015
- Wallace R.G., Twomey L.C., Custaud M.A., Moyna N., Cummins P.M., Mangone M. and Murphy R.P. Potential Diagnostic and Prognostic Biomarkers of Epigenetic Drift within the Cardiovascular Compartment. BioMed Research International 2016 Nov 24. PMID: 26942189 [impact factor 1.58]
2015
- Kotagama K., Babb C., Wolter J.M., Murphy R., and Mangone M. The human 3′UTRome v1: a clone repository for studies in post-transcriptional gene regulation. (2015) BMC Genomics 2015 Dec. 16:1036 PMID: 26645212 [impact factor 3.986]
- Kotagama K., Chang Y., and Mangone M. miRNAs as Biomarkers in Chronic Myelogenous Leukemia. Drug Dev Res. 2015 Aug 18. doi: 10.1002/ddr.21266. PMID: 26284455 [impact factor 0.73]
- Blazie S., Babb C., Wilky H., Rawls A., Park J.G. and Mangone M. Comparative RNA Seq Analysis Reveals Pervasive Tissue-specific Alternative Polyadenylation in Caenorhabditis elegans Intestine and Muscles. BMC Biology 2015 Jan. 13:4 PMID: 25601023 [impact factor 7.98]
- Wolter J.M., Kotagama K., Babb C. and Mangone M. Detection of miRNA Targets in High-Throughput Using the 3'LIFE Assay. J Vis Exp. 2015 May 25;(99): e52647 PMID: 26066857 [impact factor 1.33]
2014
- Wolter JM, Kotagama K*, Pierre–Bez A.C., Firago M, Tennant M and Mangone M. 3'LIFE: A Functional Assay to Detect C. elegans miRNA Targets in High–Throughput. Nucleic Acids Research July 29, 2014). PMID: 25074381 [impact factor 9.11]
Peer–Reviewed Publications generated from work at least partially conducted prior to ASU
- The modENCODE CONSORTIUM. Integrative Analysis of Functional Element in the Caenorhabditis elegans Genome by the modENCODE Project. Science. Dec 24, 2010; 330(6012): 1775–87 PMID: 21177976 [impact factor 32.452]
- Mangone M., Prasad Manoharan A., Thierry–Mieg D., Thierry–Mieg J., Han T., Mackowiak S., Mis E., Zegar C., Gutwein M.R., Khivansara V., Attie O., Chen K., Salehi–Ashtiani K., Vidal M., Harkins T., Bouffard P., Suzuki Y., Sugano S., Kohara Y., Rajewsky N., Piano F., Gunsalus K.C., Kim J.K. The Landscape of C. elegans 3’UTRs. Science, 2010 Jul 23; 329(5990): 432–5 PMID: 20522740 [impact factor 31.48]
- Mangone M., Myers M.P., Herr W. Role of the HCF–1 Basic Region in Sustaining Cell Proliferation. PLoS ONE. 2010; Feb 2; 5(2): e9020 PMID: 20126307 [impact factor 4.537]
- The modENCODE CONSORTIUM. Unlocking the secrets of the genomes, Nature. 2009; Jun 18; 459(7249): 927–30 PMID: 19536255 [impact factor 36.235]
- Mangone M., Macmenamin P., Zegar C., Piano F., Gunsalus KC. UTRome.org: a platform for 3'UTR biology in C. elegans. Nucleic Acids Res. 2008 Jan; 36: D57–62. Epub 2007 Nov 5 PMID: 17986455 [impact factor 9.11]
- Stein LD., Mungall C., Shu SQ., Caudy M., Mangone M., Day A., Nickerson E., Stajich J.E., Harris T., Arva A., Lewis S. The Generic Genome Browser: A Building Block for a Model Organism System Database. Genome Res., Oct 2002; 12: 1599–1610 PMID: 12368253 [impact factor 13.85] [GS]
- Piano F., Schetter A.J., Mangone M., Stein LD., Kemphues KJ. RNAi analysis of genes expressed in the ovary of Caenorhabditis elegans. PMID: 11137018 [GS] Current Biology 10: 1619–1622 2001 [impact factor 9.92]
- Stein L, Mangone M., Schwarz E., Durbin R., Thierry–Mieg J., Spieth J., Sternberg P. WormBase: network access to the genome and biology of Caenorhabditis elegans. Nucleic Acids Res. 2001 Jan 1; 29(1): 82–6 PMID: 11125056 [impact factor 7.147]
Total Number of citations: 2,006
Average number of citations per article: 125.38
Number of citations as of Sept 2014, ISI Web of Knowledge [Impact factor as of 2014]
Research Grants – Awarded and Ongoing
- NIH/NIGMS R01 GM118796-01 04/01/2016 - 03/31/2021 PI: Mangone M. (100%) Genetics and Genomics of Alternative Polyadenylation and miRNA Regulation in C. elegans.
- NIH/NCI R21 CA179144-01A1 09/12/2014 - 08/31/2017 PI: Mangone M. (90%), co-PI: Anderson (ASU) (10%) Detection and Validation of miRNA Targets in Breast Cancer
- NIH/NIGMS R01 AG050653-01A1 04/01/2016 - 03/31/2019 PI: Prahlad (U Iowa), co-PI: Mangone M. (8.6%) Uncovering How Serotonergic Signaling Non-autonomously Regulates Protein Homeostasis
- NIH/NIGMS R21-HD090707-01A1 08/01/2017 - 07/31/2019 PI: Reiner D. (Texas A&M), co-PI: Mangone M. (15%) Global changes in the 3’UTRome toggle responsiveness to growth factors
Research Grants – Awarded and Completed
- BD Competitive Bridge Funding 09/01/2014 - 09/01/2015 PI: Mangone M. (78%), co-PI: Chang (ASU) Dissecting drug resistance to Gleevec in CML patients
- ASU/DCU Catalyst Fund 08/01/2014 - 11/01/2014 PI: Mangone M. (90%), co-PI: Murphy (DCU) Expanding the human 3'UTRome library.
Courses
2025 Summer
| Course Number | Course Title |
|---|---|
| CHM 392 | Intro to Research Techniques |
2025 Spring
| Course Number | Course Title |
|---|---|
| BIO 493 | Honors Thesis |
| BCH 392 | Intro to Research Techniques |
| BCH 492 | Honors Directed Study |
| BCH 493 | Honors Thesis |
| BIO 492 | Honors Directed Study |
| CHM 392 | Intro to Research Techniques |
| CHM 492 | Honors Directed Study |
| CHM 493 | Honors Thesis |
| MBB 495 | Undergraduate Research |
| BIO 495 | Undergraduate Research |
| CHM 392 | Intro to Research Techniques |
| BCH 392 | Intro to Research Techniques |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| BCH 392 | Intro to Research Techniques |
| BIO 340 | General Genetics |
2024 Fall
| Course Number | Course Title |
|---|---|
| BCH 392 | Intro to Research Techniques |
| BCH 492 | Honors Directed Study |
| BCH 493 | Honors Thesis |
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| CHM 392 | Intro to Research Techniques |
| CHM 492 | Honors Directed Study |
| BIO 495 | Undergraduate Research |
| MBB 495 | Undergraduate Research |
| CHM 493 | Honors Thesis |
| BIO 494 | Special Topics |
| BIO 598 | Special Topics |
| BCH 392 | Intro to Research Techniques |
| CHM 392 | Intro to Research Techniques |
2024 Spring
| Course Number | Course Title |
|---|---|
| BIO 493 | Honors Thesis |
| MBB 495 | Undergraduate Research |
| BIO 495 | Undergraduate Research |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
2023 Fall
| Course Number | Course Title |
|---|---|
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| BIO 495 | Undergraduate Research |
| MBB 495 | Undergraduate Research |
| BIO 494 | Special Topics |
| BIO 598 | Special Topics |
2023 Spring
| Course Number | Course Title |
|---|---|
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| MBB 495 | Undergraduate Research |
| BIO 495 | Undergraduate Research |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
2022 Fall
| Course Number | Course Title |
|---|---|
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| BIO 495 | Undergraduate Research |
| MBB 495 | Undergraduate Research |
| BIO 494 | Special Topics |
| BIO 598 | Special Topics |
2022 Spring
| Course Number | Course Title |
|---|---|
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| MBB 495 | Undergraduate Research |
| BIO 495 | Undergraduate Research |
| BIO 340 | General Genetics |
2021 Fall
| Course Number | Course Title |
|---|---|
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| BIO 495 | Undergraduate Research |
| MBB 495 | Undergraduate Research |
| BIO 494 | Special Topics |
| BIO 598 | Special Topics |
2021 Spring
| Course Number | Course Title |
|---|---|
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| MBB 495 | Undergraduate Research |
| BIO 495 | Undergraduate Research |
| BIO 340 | General Genetics |
2020 Fall
| Course Number | Course Title |
|---|---|
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| BIO 495 | Undergraduate Research |
| MBB 495 | Undergraduate Research |
| BIO 494 | Special Topics |
| BIO 598 | Special Topics |
2020 Spring
| Course Number | Course Title |
|---|---|
| BIO 492 | Honors Directed Study |
| BIO 493 | Honors Thesis |
| MCB 701 | Seminar:Molecular/Cellular Bio |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| MBB 495 | Undergraduate Research |
| BIO 495 | Undergraduate Research |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
| BIO 340 | General Genetics |
Member of Genetics Society of America (GSA) Member of the RNA Society
Service
2011–present Member of the Executive Committee of the Molecular & Cellular Biology PhD program in the School of Life Sciences at Arizona State University.
2015–present Arizona State University Senate member (Representing the ASU School of Life Sciences).
2015–present Arizona State University Residency Appeal Committee.
Fall 2012 Co–chair of the 2012 Molecular & Cellular Biology PhD program Admission Committee.
Fall 2013-Present Member of the 2013 Molecular & Cellular Biology PhD program Admission Committee.
Ad-Hoc Review
- PLoS ONE, Manuscript Reviewer (2014 - Present)
- PLos Genetics, Manuscript Reviewer (2016-Present)
- Nucleic Acids Research, Manuscript Reviewer (2014 - Present)
- BMC Evolutionary Biology, Manuscript Reviewer (2013 - Present)
- Journal of Proteomics and Genomics Research, Manuscript Reviewer (2013 - Present)
- Journal of Proteome Research, Manuscript Reviewer (2013 - Present)
- Genes & Development, Manuscript Reviewer (2011 - Present)